FDA-Approved Radioligand Therapy For Metastatic Prostate Cancer
Advanced Treatment for Prostate Cancer with Lu-177 PSMA Therapy
Experience precision-driven cancer care with targeted Lu-177/Ac-225 PSMA therapy, designed to selectively treat PSMA-positive prostate cancer while preserving healthy tissues.
About Lu-177 PSMA (Pluvicto) Therapy
Lutetium-177 PSMA therapy is a type of targeted radioligand therapy that precisely delivers radiation to prostate cancer cells expressing PSMA (Prostate-Specific Membrane Antigen).
- PSMA ligand binds specifically to prostate cancer cells
- Lu-177 delivers targeted beta radiation
- Together, they form Pluvicto (Lu-177 PSMA-617), now FDA-approved for treating advanced prostate cancer.
FDA-Approved for metastatic prostate cancer patients, even before starting chemotherapy.
It is highly Targeted, safe, and effective therapy guided by PSMA PET imaging.
Patient Eligibility for Lu-177 PSMA Therapy
Lu-177 PSMA therapy is a targeted radioligand treatment designed for men with advanced prostate cancer who express PSMA on imaging. It is suitable for patients with progressive or metastatic disease.

Advanced Prostate Cancer
Progression After Hormonal Therapy
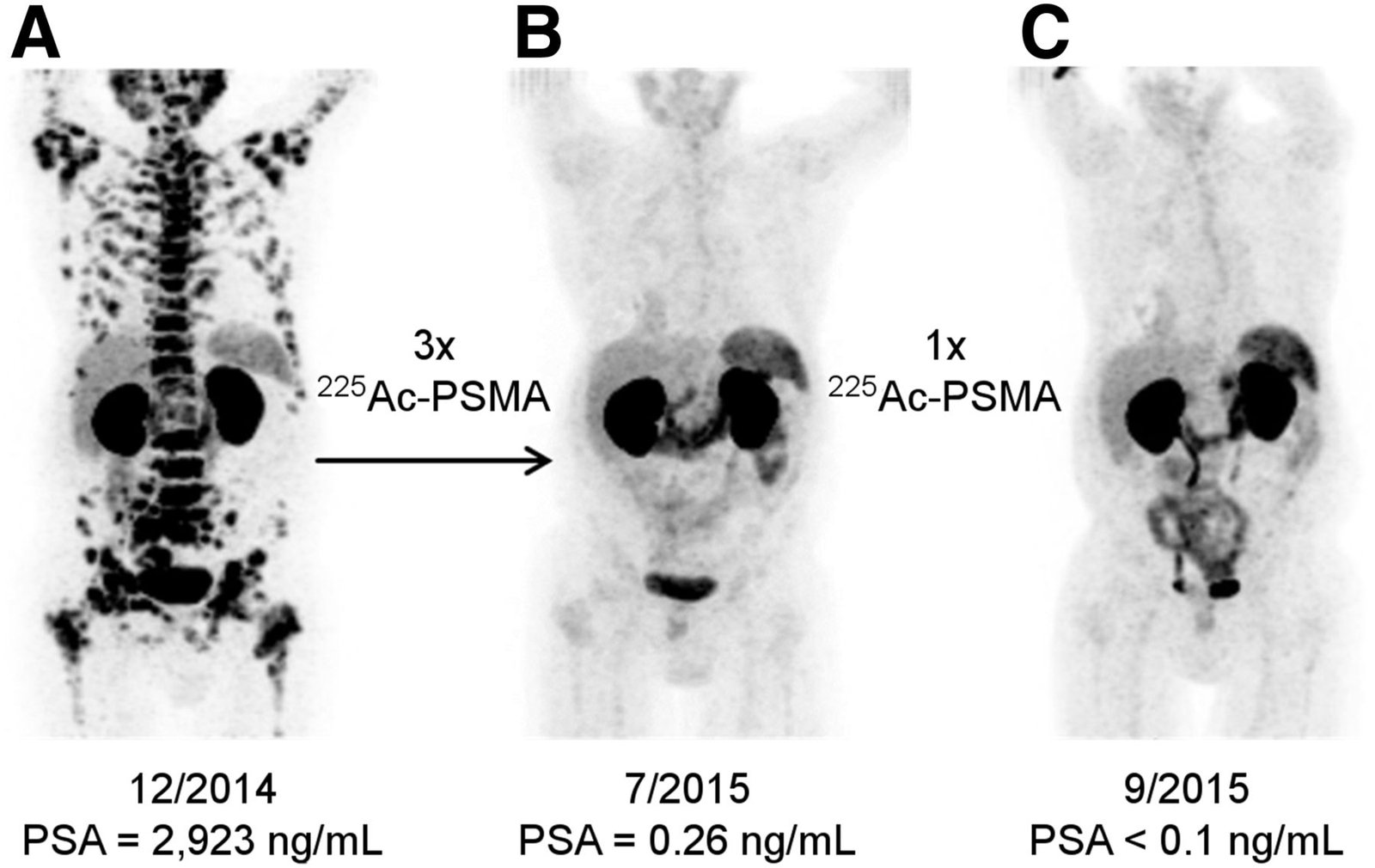
PSMA-Positive & Early Access
How Does PSMA Therapy Work
PSMA ligands selectively attach to prostate cancer cells
Lu-177 emits beta particles to kill tumor cells
The precision minimizes side effects on surrounding healthy cells
Conditions Treated with Lu-177 PSMA Therapy
01. Metastatic Castration-Resistant Prostate Cancer (mCRPC)
Advanced prostate cancer that continues to progress despite medical or surgical testosterone suppression.
02. Progressive prostate cancer not responding to hormone therapy
Disease progression even after standard hormone-based treatments fail to control tumor growth.
03. Chemotherapy-naïve mCRPC patients (FDA-approved indication)
Eligible patients who have not received chemotherapy and qualify for approved PSMA-targeted therapy.
04. Other PSMA–Positive Metastatic Tumours
Eligible for patients expressing PSMA on PET Scan like adenoid cystic carcinoma, salivary gland tumours etc.
Why Choose Our Centre for PSMA Therapy
-
Leading Theranostics Facility
We are a state-of-the-art centre specializing in advanced PSMA-targeted therapies for prostate cancer, offering cutting-edge diagnostics and treatment under one roof.
-
Expert Medical Team
Our team includes experienced nuclear medicine physicians and oncologists, ensuring precise diagnosis, personalized treatment, and expert supervision at every stage.
-
PET-Based Selection & Monitoring
Treatment decisions are guided by PSMA PET imaging, which allows accurate patient selection and precise monitoring of therapy response.
-
Premium Therapy Suites
Patients receive care in comfortable, private therapy suites with fully equipped isolation rooms, maintaining safety and privacy during treatment.
-
International Protocols with Local Affordability
Our treatments follow global safety and procedural standards while remaining accessible and affordable for Indian patients.
Frequently Asked Question
Yes. Lu-177 PSMA therapy (Pluvicto) is approved for metastatic prostate cancer and recognized by most major Indian insurance providers. Our dedicated team will assist with insurance pre-approvals and claims.
Subsribe To Our Newsletter
Stay in touch with us to get latest news and special offers.